
Clario advances decentralized clinical trials with the Power of Certainty
Decentralized clinical trials (DCT) continue to positively impact recruitment, retention and patient convenience, while expanding opportunities for diversity in clinical trials. Data integrity can still be preserved even when the responsibility for endpoint data collection moves from qualified staff to clinical study participants.
Clario’s scientific expertise, global scale and broad endpoint technology platform provide the certainty that clinical evidence in your DCT will be generated with reliability and precision.
Let us help you bring certainty to your decentralized clinical trial.
Available solutions
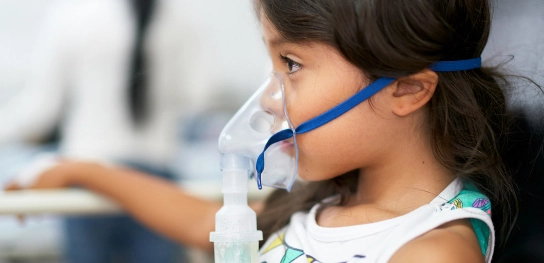
Respiratory
iSpiro
Enable patients in decentralized and hybrid clinical trials to conduct full spirometry from home with Clario iSpiro®. It combines the iSpiro app and a handheld device with an easy-to-use spirometry sensor that also meets American Thoracic Society/European Respiratory Society (ATS/ERS) requirements.
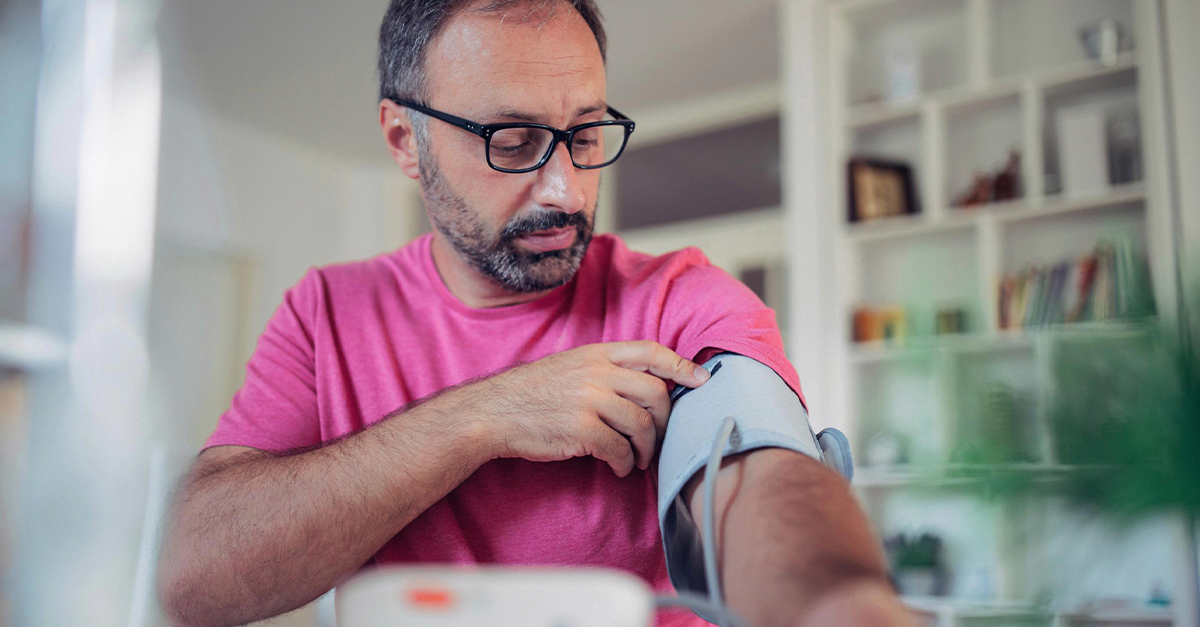
Cardiac Safety
Home Blood Pressure
Collect high-quality blood pressure endpoint data.
Patient-acquired ECG
Drive cardiac safety for your DCT with rigorous, responsive and easy to use patient-administered ECGs from home.

eCOA
Enable patients to participate in your decentralized clinical trial wherever they are through a choice of flexible technologies.
- Telehealth

Precision Motion
Opal® V2C System
Capture digital mobility outcomes with medical-grade wearable sensors, pre-configured validated assessments and an analytics platform that all provide flexible options for PerfO Active Assessments or Passive Remote Patient Monitoring for your DCT.

Medical Imaging
Clario supports clinical trials from inception to post-trial assessments. The powerful combination of our medical and scientific experts and technology solutions delivers superior trial management and insight at every stage.
Decentralized and hybrid clinical trial experience and expertise
20
years of deploying DCT solutions globally
200+
endpoint and therapeutic area experts
100+
languages supported
3
continents with device, provisioning and logistics hubs
24/7
patient, site and sponsor support
Decentralized clinical trials: What does certainty mean for clinical development?
Endpoint and therapeutic area expertise reduce
site and patient burden
- Simplify site and patient experiences for your decentralized clinical trials with Clario’s science-driven strategies.
- Deliver rich, regulatory-compliant data with support from Clario’s 200+ endpoint science and therapeutic area experts, who can advise on appropriate clinical outcome assessments for your hybrid or virtual trial.
Global coverage and local expertise in assessing drug quality and safety for all
- Scale your DCT globally and reach diverse and remote patient populations.
- Know that the right devices — properly configured — are sent to the right sites and patients thanks to Clario’s vast network of logistic hubs across Europe, Asia and the Americas.
- Evidence a product’s safety and efficacy with certainty by leveraging Clario’s worldwide capabilities — all backed by 50 years of global regulatory experience.
Broad endpoint technology platform simplifies the DCT experience
- Quickly capture, manage and analyze data for all clinically meaningful endpoints across all major therapeutic areas.
- Access all of Clario’s decentralized and hybrid evidence generation solutions for cardiac safety, medical imaging, eCOA, respiratory and precision motion via one centralized platform.
How does a broad endpoint technology platform benefit your DCT?
- Improves efficiency in DCT start-up while maintaining consistency and speed in data management and delivery.
- Simplifies the user experience for patients, sites and sponsors.
- Enhances coordination across data capture modalities.
- Paves the way for unlocking composite endpoints and outcome measures that can lead to clinical trials that need fewer patients and are lower cost—all while accelerating timelines.
Better patient experience for better endpoint data
Our hybrid and decentralized clinical trial software solutions and connected devices are designed with patients in mind. They can be comfortably and effectively used from the comfort of home or just about anywhere for more certain outcomes.
Our patient-centric approach includes:
Device and solution training
Guided workflows with smart reminders and notifications
Multilanguage capability
24/7 patient support
Talk to a specialist
Our team of experts is available to address any questions you may have. Submit your contact information and we’ll be in touch shortly.
